Introduction
Lumbar instability (LI) is classified as a type of mechanical low back pain (LBP) and has been the focus of numerous studies investigating its diagnosis, prevalence, risk factors, involvement of other structural impairments, and effective treatments and exercises [1–7]. This interest may be due to its frequent association with the current LBP, which can negatively affect work productivity and quality of life [2, 5]. The reported prevalence of LI ranges from 13% to 75%, based on physical examinations, x-ray imaging, and lumbar instability questionnaires [5, 8, 9].
Classical theory suggests that LI arises from dys-function in at least one of three subsystems: the passive subsystem, the active subsystem, and the neural control subsystem [10]. Traditionally, diagnosis has relied on radiographic assessment, which primarily reflects impairments in the passive subsystem [11, 12]. However, recent studies have increasingly emphasised the roles of the active and neural control subsystems [13, 14]. The active subsystem includes muscles that contribute to spinal stability, such as the abdominal and back muscles, pelvic floor muscles, and respiratory muscles [15].
Trunk muscle function in individuals with LI associated with disc degeneration, as detected by magnetic resonance imaging, has been extensively studied. Silfies et al. [16] used electromyography during a standing reach test and found that LI patients exhibited increased activation of superficial trunk muscles (external oblique and rectus abdominis) and a reduced deep-to-superficial muscle activation ratio (internal oblique versus rectus abdominis), indicating altered neuromuscular control. Later, Silfies et al. [13] reported delayed activation of the multifidus and erector spinae muscles in LI patients during a self-initiated arm perturbation task. Similarly, Davarian et al. [14] observed reduced trunk extensor strength and endurance in individuals with LI compared to those with CLBP. Collectively, these findings suggest that LI is characterised by reduced trunk muscle strength, delayed activation of deep trunk muscles, and increased activation of superficial trunk muscles, which may disrupt normal spinal movement patterns and contribute to symptoms during physical activity [13–17].
Respiratory muscles, particularly the diaphragm, also contribute to trunk stability. Beyond their primary role in respiration, the diaphragm and associated respiratory muscles stabilise the spine by generating intra-abdominal pressure [15, 18]. Evidence regarding respiratory muscle function in individuals with LBP remains inconsistent. Mohan et al. [19] reported no significant differences in respiratory muscle strength or endurance compared to healthy controls, whereas Shah et al. [20] observed reduced respiratory muscle endurance in LBP patients relative to healthy subjects. Similarly, Janssens et al. [21], using inspiratory muscle loading protocols, demonstrated more rapid fatigue of the diaphragm and other inspiratory muscles in individuals with LBP. These discrepancies may reflect methodological differences or the heterogeneity of CLBP populations.
Notably, Bunphrom et al. [22] investigated diaphragm excursion and thickness using real-time ultrasound imaging in patients with CLBP and LI. They reported reductions in both diaphragm excursion and thickness in LI patients. To date, this remains the only study confirming such impairments in this population, highlighting the need for further investigation.
Although numerous studies have investigated respiratory muscle function in CLBP patients compared to healthy individuals [20, 21], many have interpreted their finding through the lens of LI, even when the participants were broadly classified as having CLBP. Moreover, existing research has primarily focused on the function of back and abdominal muscles in patients with LI. Despite the crucial role of respiratory muscles in trunk stability, their strength and endurance have not been specifically examined in this population.
Therefore, assessing the strength and endurance of respiratory muscles is essential for informing management strategies in patients with LI. Understanding how these parameters differ among individuals with CLBP with and without LI, as well as healthy individuals, is crucial. Accordingly, the present study aimed to compare the strength and endurance of respiratory muscles among these three groups.
We hypothesised that individuals with LI would exhibit significantly lower respiratory muscle strength and endurance compared to CLBP and healthy groups. The findings are expected to provide valuable insights for optimising treatment approaches and developing specific recommendations for this population.
Material and methods
Study design
The study took place at the BNOJPH Laboratory, Faculty of Associated Medical Sciences, Khon Kaen University, between September 2023 and March 2025. All participants were informed of the study protocol and provided written informed consent prior to participation.
Participants
Participants included healthy individuals and individuals with CLBP. The sample size was calculated by comparing means across more than two groups [23], using parameters derived from a pilot study with 22 participants per group. The formula: sample size = was used, where = 2.41 for a three-group comparison, = 0.84 for 80% power, = 22.01, and = 15.98. Based on these values, the required sample size was 37 participants per group.
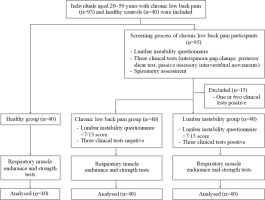
Inclusion criteria were: aged 20–59 years, LBP lasting more than 3 months with a pain intensity score of 3 to 7 on the visual analogue scale (VAS) [24], and healthy individuals with no history of LBP in the past six months. Exclusion criteria included pregnancy, body mass index (BMI) greater than 30 kg/m2, chronic respiratory diseases (e.g., chronic obstructive pulmonary disease, asthma, pulmonary hypertension), neuro-muscular disorders (e.g., peripheral neuropathy, spinal cord injury, myopathy, motor neuron disease), history of pelvic injury, or severe pathological spinal conditions (e.g., scoliosis or spondylolisthesis). Participants reporting a pain score greater than 7 on the VAS during testing were withdrawn from the study. Figure 1 presents a flow diagram of the participant progression throughout the study.
Group classification and screening process
Participants with no history of LBP in the past six months were classified as the healthy group. Participants with CLBP were further divided into two groups: the CLBP group and the LI group, based on the lumbar instability questionnaire and three clinical tests. Thresholds for these assessments were determined via x-ray imaging. The lumbar instability questionnaire demonstrated a sensitivity of 100% and a specificity of 26% [2]. A positive result on all three clinical tests yielded a 67% probability of identifying LI in CLBP patients [3].
CLBP who scored 7/14 on the lumbar instability questionnaire [2] and showed positive results on all three clinical tests (interspinous gap change during flexion-extension, passive accessory intervertebral movement tests (PAIVMs), and posterior shear test) [3] were classified into the LI group. Participants scoring < 7/14 and not positive on all three clinical tests were classified into the CLBP group. The group classification was performed by researcher AL, who was not involved in outcome measurements.
Pulmonary function was assessed using spirometry (VyntusTM SPIRO, Vyaire Medical, Germany) to exclude participants with pulmonary diseases. Researcher SP conducted the assessment using the forced vital capacity (FVC) manoeuvre in accordance with the American Thoracic Society (ATS) and European Respiratory Society (ERS) guidelines [25]. Participants’ details (age, height, sex, and ethnicity) were entered into the spirometer’s software. The spirometry software automatically generated FVC, forced expiratory volume in the first second (FEV1), FEV1/FVC, and peak expiratory flow (PEF), which were interpreted using Thai-specific reference values [26]. If all three trials satisfy the predefined acceptability and reproducibility criteria, and the best-performing trial was selected for subsequent analysis.
Outcome measurement
The overall respiratory muscles, including the diaphragm, intercostal muscles, abdominal muscles, and accessory muscles of respiration, were assessed for both strength and endurance [27]. Maximal inspiratory pressure (MIP), maximal expiratory pressure (MEP), and maximal voluntary ventilation (MVV) were reported as observed values rather than predicted values because participants did not have pulmonary disorders.
Respiratory muscle strength
Respiratory muscle strength was measured using a respiratory pressure meter (MicroRPM, Micro Medical, United Kingdom), recorded in cm H2O. During the MIP and MEP assessment, participants were instructed to hold the device with both hands, seal their lips around the mouthpiece, and wear a nose clip to prevent air leakage.
For the MIP measurement, participants exhaled fully to residual volume (RV) before inhaling forcefully for 1 s against the device’s resistance. For the MEP measurement, participants inhaled fully to total lung capacity (TLC) and then exhaled forcefully for 1 s against the device. Each measurement was repeated three times, ensuring values differed by no more than 5% [28]. The highest value was recorded for further analysis. Testing was performed by researcher PT, who was blinded to the group allocation.
Respiratory muscle endurance
Endurance of the respiratory muscles was assessed via MVV using spirometry (VyntusTM SPIRO, Vyaire Medical, Germany). Participants breathed as deeply and rapidly as possible for 12 s following ATS guidelines [25]. MVV value was expressed in litres per minute (l/min). Testing was conducted by researcher SP, who was blinded to the participants’ group allocation.
Statistical analysis
Data were analysed using SPSS version 29. Baseline characteristics of the participants were presented as mean values with standard deviations for the continuous variables and as frequencies and percentages for the categorical variables. The Shapiro–Wilk test was selected to assess the normality of the data. Differences in respiratory muscle strength and endurance among the three groups were analysed using one-way ANOVA with Least Significant Difference (LSD) post-hoc tests. A p-value of less than 0.05 was considered statistically significant.
Results
Baseline characteristics
Table 1 summarises the baseline characteristics of the participants across the three groups. A statistically significant difference in pain score, measured by VAS, was observed between the CLBP group and the LI group (p-value < 0.001). No significant difference was found in pain duration (months) between these two groups. Participants in all three groups reported various underlying conditions, including allergies, hypertension, dyslipidemia, and diabetes mellitus. One participant in the CLBP group also reported a history of systemic lupus erythematosus (SLE).
Table 1
Participant baseline characteristics (value represents number shown as percentage unless otherwise indicated)
Regarding lung function, no participants reported any pulmonary disease and there were no statistically significant differences in lung function among the three groups.
Comparison of respiratory muscle strength and endurance
Comparison of respiratory muscle strength revealed statistically significant differences in MIP between the healthy and LI group (p-value < 0.001), as well as between the CLBP and LI group (p-value < 0.04). For respiratory muscle endurance, measured by MVV, a statistically significant difference was observed between the healthy and LI group (p-value = 0.007). These findings are summarised in Table 2 and illustrated in Figure 2.
Figure 2
Comparison of respiratory muscle strength and respiratory muscle endurance among the three groups
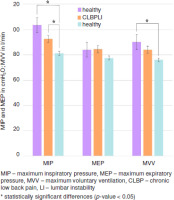
Table 2
Comparison of respiratory muscle strength and respiratory muscle endurance for the three groups
[i] MIP – maximum inspiratory pressure, MEP – maximum expiratory pressure, MVV – maximum voluntary ventilation, CLBP – chronic low back pain, LI – lumbar instability
* p-value < 0.05, ** p-value < 0.001
a statistically different from healthy group, b statistically different from CLBP group, c statistically different from healthy group
Discussion
This study is the first to compare respiratory muscle strength and endurance among individuals with CLBP, those with LI, and healthy individuals. The findings revealed that participants with LI had significantly lower inspiratory muscle strength compared to both those with CLBP and healthy participants. Additionally, LI participants exhibited lower respiratory muscle endurance than healthy individuals.
Participants in this study included healthy individuals with no history of LBP in the past six months, individuals with CLBP lasting at least three months, and individuals with LI. Participant ages were controlled across groups, as age is known to influence degenerative changes in the lumbar spine [5]. Baseline analysis revealed a significant difference in pain intensity between the CLBP and LI groups, but no significant difference in pain duration. Pulmonary function tests showed no evidence of respiratory impairment in any group, with an average FEV1/FVC ratio of 88.51% [25].
The significant reduction in inspiratory muscle strength observed in LI participants may be explained by altered trunk muscle activation patterns associated with LI. These include delayed activation of deep trunk muscles [13], a decreased ratio of deep to superficial abdominal muscle activity [16], and reduced back muscle strength [14], which are more pronounced in LI than in general CLBP. Our finding suggests that inspiratory muscle weakness in LI reflects broader impairments in core muscle function. This aligns with previous reports that postural changes such as transitioning from sitting to standing or turning while supine often exacerbate pain in LI patients due to insufficient intra-abdominal pressure generation [2]. Consequently, the LI group in our study demonstrated significantly higher pain scores than the CLBP group, likely due to the cumulative stress on the lumbar spinal segment from inadequate core muscle function.
Our results are supported by previous studies examining the role of inspiratory muscle strength in CLBP management. Janssens et al. [29] reported that high-intensity inspiratory muscle training improved respiratory muscle strength and reduced pain intensity in individuals with CLBP. Similarly, Vicente-Campos et al. [30] demonstrated that an eight-week progressive abdominal gymnastics exercise program increased diaphragm thickness and MIP, while decreasing disability and pain scores in patients with CLBP.
Although the mean inspiratory muscle strength in the CLBP group was lower than in the healthy individuals, this difference was not statistically significant, consistent with previous studies reporting similar respiratory muscle strength between healthy participants and those with CLBP [19, 20].
Regarding respiratory muscle endurance, the LI participants showed significantly lower MVV compared to the healthy group, which is consistent with the findings by Shah et al. [20] in patients with CLBP. As previously noted, impairments in LI involve multiple core muscle groups that are functionally interconnected and act synergistically to maintain trunk stability [31]. Reduced strength in these muscles likely compromises overall endurance and increases the susceptibility to fatigue. While the CLBP group exhibited slightly lower endurance than the healthy group, this difference did not reach statistical significance.
This study provides valuable clinical insights; however, some limitations should be noted. First, although our outcome assessment followed ATS and ERS protocols, test–retest reliability was not evaluated, limiting confidence in rater consistency. Second, participants were not stratified by sex at enrolment, resulting in a higher proportion of female participants; therefore, the finding may primarily reflect female responses and limit the generalisability to males. Third, the reliability of the three clinical tests used for group allocation was not assessed, reducing confidence in the reproducibility of group classification. Finally, the cross-sectional design limits the ability to infer causal relationships.
Conclusions
Our findings partially support the study hypothesis, as patients with LI exhibited lower inspiratory muscle strength compared to both healthy individuals and those with CLBP. Respiratory muscle endurance was also reduced in patients with LI relative to healthy controls. These results highlight that patients with LI exhibit greater respiratory muscle weakness than those with CLBP, underscoring the need for interventions that emphasise core muscle training, particularly targeting the respiratory muscles. Such interventions may improve trunk stability and potentially prevent further structural compromise.